History is littered with the corpses of ‘settled’ medical truths. Between 1860 and 1920, the medical establishment championed cocaine as a panacea for everything from hay fever to nerve exhaustion. Even more striking is the 70-year window between the late 1800s and 1964, during which physicians recommended specific cigarette brands for ‘throat protection’ and lung health. It took seven decades of mounting evidence before the Surgeon General finally shattered that envelope of medical denial.
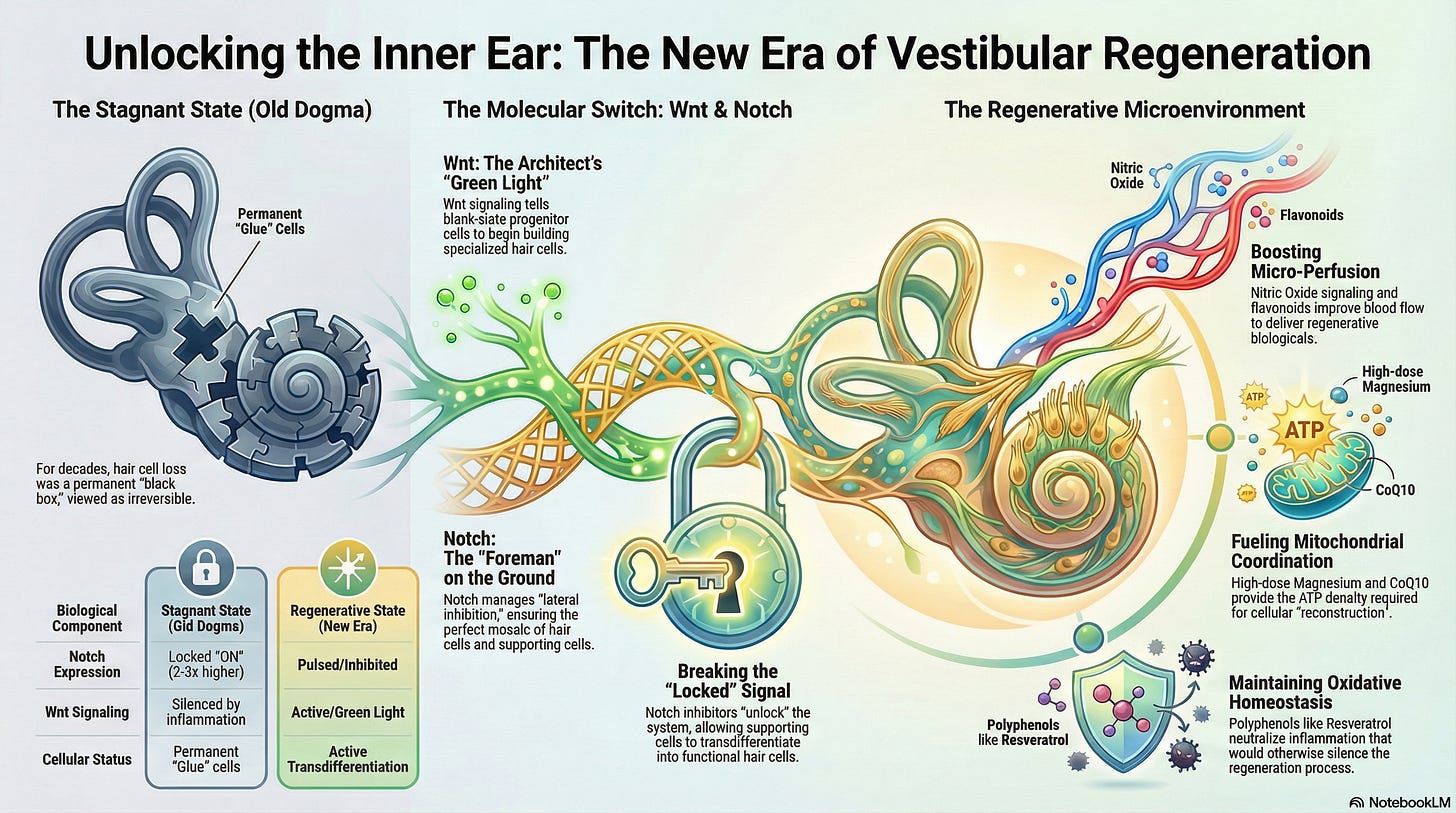
Today, vestibular medicine is trapped in a similar state of ‘settled’ stagnation. For over fifty years, the prevailing dogma insisted that because mammalian hair cells do not spontaneously regenerate, any damage is permanent. We have treated the inner ear as a ‘black box’, a region of biological finality. We told our patients they must simply ‘cope.’ However, in 2026, groundbreaking research overturned this notion, revealing that the inner ear is not a dead end; it is a programmable landscape waiting for the correct ‘signaling.’ One pivotal study demonstrated that a specific combination of molecular signals can activate regeneration pathways, allowing previously damaged hair cells to recover their function, marking a significant shift in our understanding of auditory and vestibular healing.
THE MOLECULAR ARCHITECTS: WNT AND NOTCH
To move past ‘therapeutic nihilism,’ we must understand the two specific proteins that act as the master architects of the balance system. Specifically, in the context of cellular biology, every supporting cell in the inner ear functions akin to precursor or progenitor cells, performing tasks as ‘General Contractors’ waiting for specific orders from these proteins to initiate their cellular functions.
THE WNT SIGNAL: THE ARCHITECT’S BLUEPRINT
Wnt is a family of secreted glycoproteins that acts as the ‘Green Light’ for growth and identity.
The Role: Wnt signaling instructs a generic progenitor cell to cease being a ‘blank slate’ and initiate differentiation into a specialized hair cell (Atkinson et al., 2015).
The Impact: It activates the master transcription factor ‘Atoh1’, which is the blueprint for building stereocilia. Without Wnt, the inner ear is still a silent, empty room.
THE NOTCH SIGNAL: THE SITE FOREMAN
Notch is a ‘contact-dependent’ signaling system. If Wnt is the architect with the big idea, Notch is the foreman on the ground, preventing ‘over-building.’
The Role (Lateral Inhibition): When a cell begins to differentiate into a hair cell, it uses Notch to ‘touch’ its neighbors. It tells them: ‘I am the hair cell. You must remain a Supporting Cell (the glue).’ This precise cellular choreography is crucial for maintaining the balance and function of the vestibular system, as it ensures the ideal ratio of hair cells to supporting cells. Studies show that preserving this mosaic through lateral inhibition can significantly improve patient outcomes by reducing dizziness scores and facilitating more effective balance recovery (Kwan et al., 2009).
The Result: This maintains a perfect mosaic—one hair cell surrounded by supporting cells to maintain systemic ‘integrity.’
BREAKING THE STUCK SIGNAL
We believed the inner ear was a ‘black box’ because, in the adult vestibular system, Notch (Red Light) is ‘locked’ in the ON position. It constantly signals to surrounding cells to remain as ‘glue,’ preventing them from ‘fixing’ the system. Studies have shown that Notch receptor expression in this state can be upregulated by nearly 2- to 3-fold, further confirming its dominance in maintaining the current cellular state (Schmeiser & Kiernan, 2014).
The breakthrough of the current era is the ability to ‘facilitate’ a change by using ‘Notch Inhibitors.’ By telling the Foreman (Notch) to take a break, the Wnt blueprint can finally be reread (Li et al., 2015). This allows supporting cells to transdifferentiate, turning from ‘glue’ into functional ‘hair cells.’ While these inhibitors have shown promising results in preclinical studies, including those conducted in animal models, they have yet to be tested in human clinical trials. We are finally ‘coordinating’ the signals to unlock the body’s own blueprints.
OPTIMIZING THE METABOLIC MICROENVIRONMENT
The transition from a state of deficit to a state of functional ‘integrity’ requires a pristine metabolic landscape. To ‘facilitate’ recovery, we must address three pillars of the vestibular ‘microenvironment.’
1. MICRO-PERFUSION AND NITRIC OXIDE SIGNALING
The labyrinthine artery is a terminal branch with no collateral circulation, making the inner ear highly susceptible to ischemic insult.
Mechanism: Utilizing compounds that ‘support’ Nitric Oxide (NO) signaling is critical. NO acts as a potent vasodilator in the inner ear microvasculature.
Clinical Insight: Flavonoids increase vestibular blood flow by reducing blood viscosity while improving NO bioavailability (Müller & Barr-Gillespie, 2015). This ‘supports’ the delivery of regenerative Biologicals to the target tissue.
2. MITOCHONDRIAL COORDINATION AND ATP DENSITY
Regeneration is an energetically expensive process. The vestibular dark cells must maintain the endolymphatic potential via the Na+/K+-ATPase pump, which consumes substantial ATP.
Mechanism: High-dose ‘Magnesium’ acts as a calcium channel antagonist and a necessary cofactor for all ATP-binding reactions.
Clinical Insight: When Magnesium levels are optimized, it prevents the metabolic ‘short-circuiting’ of hair cells. Furthermore, ‘Coenzyme Q10’ (CoQ10) ‘coordinates’ the electron transport chain, reducing the electron loss that leads to mitochondrial decay (Stojanovska & Zine, 2021).
3. OXIDATIVE HOMEOSTASIS AND CELLULAR STABILITY
A damaged inner ear is often a pro-inflammatory environment. Chronic inflammation silences the very Wnt signaling we are trying to activate.
Mechanism: We use targeted nutritional signaling to neutralize Reactive Oxygen Species (ROS) that trigger apoptotic pathways.
Clinical Insight: Polyphenols like ‘Resveratrol’ activate the SIRT1 pathway, which ‘facilitates’ cellular survival and protects the ‘integrity’ of the vestibular ganglion neurons (Schmeiser & Kiernan, 2014).
CLINICAL TRANSLATION AND BARRIERS
While the 2026 pivotal study used high-fidelity human inner-ear organoids and transgenic murine models to visualize real-time transdifferentiation, several barriers to widespread clinical translation remain. The primary challenge involves targeted delivery across the blood-labyrinth barrier without disrupting endolymphatic homeostasis. Furthermore, the manipulation of Wnt and Notch must be precisely ‘pulsed’ to avoid hyperplasia or the loss of structural ‘integrity’.
Patient selection is critical. Those with acute vestibular insult or chronic hypofunction—specifically those struggling with visual or surface dependency—are most likely to benefit as we ‘optimize’ their peripheral input. The long-term durability of these regenerated cells depends on successful synaptogenesis with the vestibular nerve, a process that requires a sustained, healthy ‘microenvironment.’
FROM SUPPRESSION TO SYSTEMIC INTEGRITY
The 19th-century medical community used cocaine and tobacco to mask the body’s distress signals. For the last fifty years, we have accepted vestibular symptoms as inevitable. In 2026, we enter the era of Biological Facilitation. By optimizing the vascular supply and coordinating the mitochondrial output, we move from the era of compensating for failure to the era of fueling success. The inner ear is no longer a black box; it is a dynamic system that we can finally begin to optimize (Staecker et al., 2014).
Moving forward, our research must prioritize several avenues. Firstly, targeted delivery methods across the blood-labyrinth barrier without disrupting endolymphatic homeostasis must be refined. Secondly, understanding the long-term effects and sustainability of regenerated hair cells necessitates further study. Thirdly, developing diagnostic tools for accurately identifying patients who stand to benefit most from these therapies is crucial. Finally, interdisciplinary collaboration will be key to integrating genetic, molecular, and engineering insights to advance our understanding and applications of these groundbreaking findings.
REFERENCES
Atkinson, P. J., Huarcaya Najarro, E., Sayyid, Z. N., & Cheng, A. G. (2015). Making sense of Wnt signaling—linking hair cell regeneration to development. Frontiers in Cellular Neuroscience, 9, 66. https://doi.org/10.3389/fncel.2015.00066
Kwan, K. Y., Brooke, R. E., Wei, W., & Corey, D. P. (2009). Atoh1 and Notch signaling in the development and regeneration of the inner ear. Annals of the New York Academy of Sciences, 1170(1), 28–33. https://doi.org/10.1111/j.1749-6632.2009.04493.x
Li, W., Wu, J., Yang, J., Sun, S., Chai, R., Chen, Z. Y., & Li, H. (2015). Notch inhibition induces mitotically generated hair cells in mammalian cochleae via activating the Wnt pathway. Proceedings of the National Academy of Sciences (PNAS), 112(1), 166–171. https://doi.org/10.1073/pnas.1415901112
Müller, U., & Barr-Gillespie, P. G. (2015). New treatment strategies for hearing loss. Science, 348(6234), 537–542. https://doi.org/10.1126/science.aaa3414
Schmeiser, S., & Kiernan, A. E. (2014). Notch signaling in mammalian hair cell regeneration. Madame Curie Bioscience Database. https://www.ncbi.nlm.nih.gov/books/NBK6176/
Staecker, H., Praetorius, M., & Brough, D. E. (2014). Optimizing Atoh1-induced vestibular hair cell regeneration. The Laryngoscope, 124(S1), S1–S12. https://doi.org/10.1002/lary.24938
Stojanovska, M., & Zine, A. (2021). Understanding the differentiation and epigenetics of cochlear sensory progenitors in pursuit of regeneration. Current Opinion in Otolaryngology & Head and Neck Surgery, 29(5), 374–380. https://doi.org/10.1097/MOO.0000000000000746