Deconstructing the ‘Central’ Dogma: The Peripheral Masquerade of Downbeat Nystagmus

As vestibular professionals, we have long accepted the clinical dogma that spontaneous Downbeat Nystagmus (sDBN) is a ‘pathognomonic hallmark‘ of central vestibulopathy. We are conditioned to immediately localize this to the vestibulocerebellum—specifically the flocculus, paraflocculus, and the nodulus/uvula complex—to explain the pathological upward slow-phase drift. However, emerging evidence from the last 24 months is forcing a radical reassessment. New data suggest that the ‘integrity of the vertical VOR microenvironment‘ can be disrupted at the peripheral level, masquerading as a central lesion through a sophisticated mechanism of vertical tonic imbalance and ‘tonic disinhibition‘.
The Foundation: Innate Vertical Reflex Asymmetry
To understand how a peripheral deficit can drive a ‘central’ sign, we must first analyze the inherent, built-in asymmetry of the vertical system. The vertical VOR is not a balanced 1:1 system. Humans possess an innate preponderance for upward eye responses—a biological ‘anti-gravity strategy‘ designed to counteract the gravitational forces that naturally oppose upward head and body movement.
Marcelli et al. (2024) elucidate the ‘anatomical drivers‘ of this bias: the Anterior Semicircular Canals (ASC) are physically positioned much closer to the sagittal (front-to-back) plane of the skull than the Posterior Semicircular Canals (PSC).
This geometric alignment creates a ‘functional preponderance‘. When the head moves in the sagittal plane, the endolymphatic flow in the ASC is caught much more efficiently than in the PSC. Consequently, the excitatory signal to move the eyes upward (driven by the ASC) is naturally ‘louder‘ and more robust than the signal to move the eyes downward (driven by the PSC). In a healthy state, this imbalance is managed by the cerebellar flocculus, which acts as a constant, tonic ‘brake‘ on the ASC’s dominant signal to maintain vertical stability.
Tonic Disinhibition: The Loss of Opposing Signals
DBN is the clinical manifestation of ‘tonic disinhibition‘—the release of that cerebellar brake. Traditionally, we localized this failure exclusively to the cerebellum or brainstem. However, Zhang et al. (2024) have provided ‘experimental-only‘ data showing that the same result occurs when the ‘opposing peripheral signal‘—the input from the PSC—is diminished or lost.
In their retrospective analysis of 54 sDBN patients, 22.2% of cases were definitively peripheral in origin, including Meniere’s Disease and isolated vertical canal hypofunction. When the PSC is hypofunctional, its weaker downward signal disappears entirely. This leaves the ‘naturally dominant‘ ASC signal unopposed. Even with a healthy cerebellar brake, the ‘signaling environment‘ is so heavily skewed that the brain receives a constant, false message of downward head rotation. The result is a persistent, slow, upward drift, which the CNS corrects with downward saccades—producing a DBN driven by the ear rather than the brain.
The Mechanics of Cross-Coupling: Signal ‘Leakage’ and the Nodulus
A frequent clinical observation is the emergence of DBN specifically after a horizontal head-shake test. This is the ‘cross-coupling‘ phenomenon, which Marcelli et al. (2024) identify as a disruption of the Nodulus and Uvula (NOD) circuits.
The NOD is responsible for ‘coordinating‘ and ‘integrating‘ signals from the semicircular canals and the otoliths to maintain spatial orientation. During a horizontal head-shake, the ‘velocity storage mechanism‘ in the brainstem is charged with energy. In a healthy system, this energy is contained and discharged strictly within the horizontal plane; however, if there is a pre-existing vertical ‘tonic imbalance‘ or a breakdown in the ‘integrity of the NOD integration‘, this energy ‘leaks‘ or ‘cross-couples‘ into the vertical plane.
This ‘perverted nystagmus‘ is the clinical proof of a system that can no longer separate horizontal and vertical planes of movement. The horizontal stimulus ‘facilitates‘ the naturally dominant upward bias of the ASC pathway, resulting in a post-head-shake DBN.
The Clinical Trap: Why Goggles Aren’t a Diagnosis
Clinicians often observe DBN that is suppressed in the light but becomes ‘violent‘ under infrared video goggles or after a horizontal head-shake. While this robust visual fixation suppression suggests the ‘cerebellar brake‘ is functioning (as a truly damaged cerebellum often cannot suppress nystagmus even in the light), it is not a ‘slam dunk’ for peripheral pathology.
Educators and clinicians must remain cautious. This new knowledge is crucial not because it replaces central localization, but because it ‘facilitates‘ a more nuanced differential diagnosis. We must ‘support‘ the peripheral hypothesis by correlating it with a constellation of ‘corresponding factors‘ to ensure we aren’t missing a ‘neuro-degenerative process‘:
vHIT Gain Asymmetry: You must look for a measurable, statistically significant decrease in PSC gain relative to the ASC in the same conjugate plane. Without a gain deficit, the peripheral theory lacks ‘integrity‘.
Otolithic Signaling: Asymmetric vertical tone is often modulated by the otoliths. Abnormal oVEMP or a tilt in the Subjective Visual Vertical (SVV) suggests a ‘sensory mismatch‘ in the gravity-sensing pathways that the Nodulus/Uvula cannot ‘coordinate‘.
Smooth Pursuit Integrity: The ‘central gatekeeper‘ (the flocculus) manages both VOR inhibition and smooth pursuit. If vertical smooth pursuit remains high-gain and smooth, it ‘supports‘ a healthy central integrator, pointing the finger back at the periphery.
Positional ‘Leakage’: Peripheral DBN is significantly more likely to manifest horizontal or torsional components during positional testing (91.7%). Pure, unadulterated vertical drift in all positions—the ‘pure vertical’—remains a major central ‘red flag‘.
Conclusion: A Blended Diagnostic Strategy
This shift in understanding demands that we move away from ‘automatic’ assumptions. DBN in the dark or after a head-shake may hide subtle central causes or represent the early ‘signaling‘ of a central breakdown. Our role is to ‘optimize the signaling environment‘ through exhaustive testing. By ‘coordinating‘ these findings—vHIT gain, VEMP symmetry, and oculomotor integrity—we avoid the ‘maladaptive strategy‘ of premature conclusions. This sophisticated approach supports a truly targeted rehabilitation plan that addresses the source of the tonic imbalance.
APA Citations
Marcelli, V., Giannoni, B., Volpe, G., Faralli, M., Fetoni, A. R., & Pettorossi, V. E. (2024). Downbeat nystagmus: a clinical and pathophysiological review. Frontiers in Neurology, 15, 1394859. https://doi.org/10.3389/fneur.2024.1394859
Zhang, S., Lang, Y., Wang, W., Wu, Y., Yan, S., Zhang, T., ... & Gu, P. (2024). Analysis of etiology and clinical features of spontaneous downbeat nystagmus: a retrospective study. Frontiers in Neurology, 15, 1326879. https://doi.org/10.3389/fneur.2024.1326879



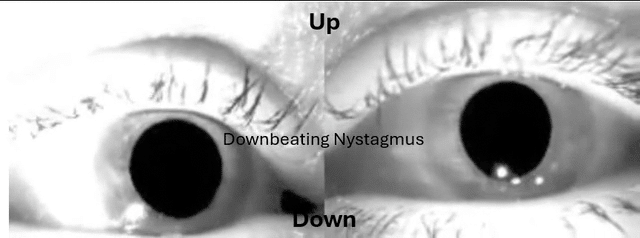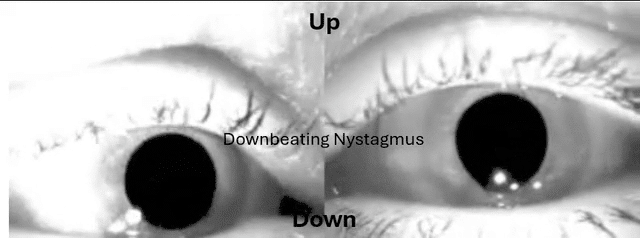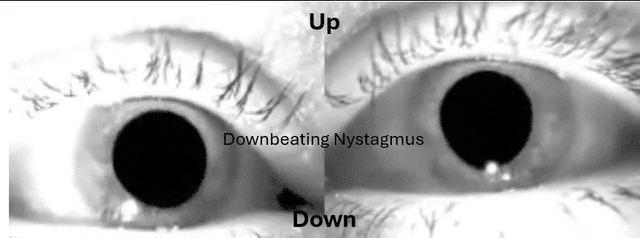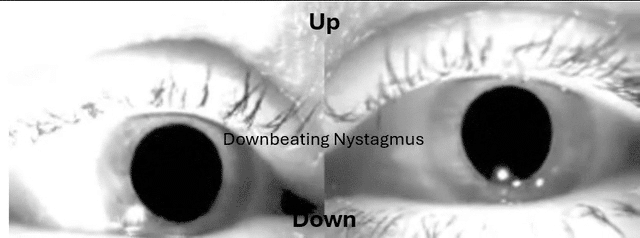
This is something many have sent me over the years and wasn't buying it until had a patient recently with utricular and saccular issues and it was the Audiologist that was on top of it.
Great job on this one Brian! I attempted to get through the 2024 article…your summary and conclusions are extremely helpful! Keep up the great work 👍